Clinical problem
ADR evidence is often scattered across messages, laboratory abnormalities, medication orders, and patient narratives. Manual reporting is slow and easy to miss during routine clinical work.
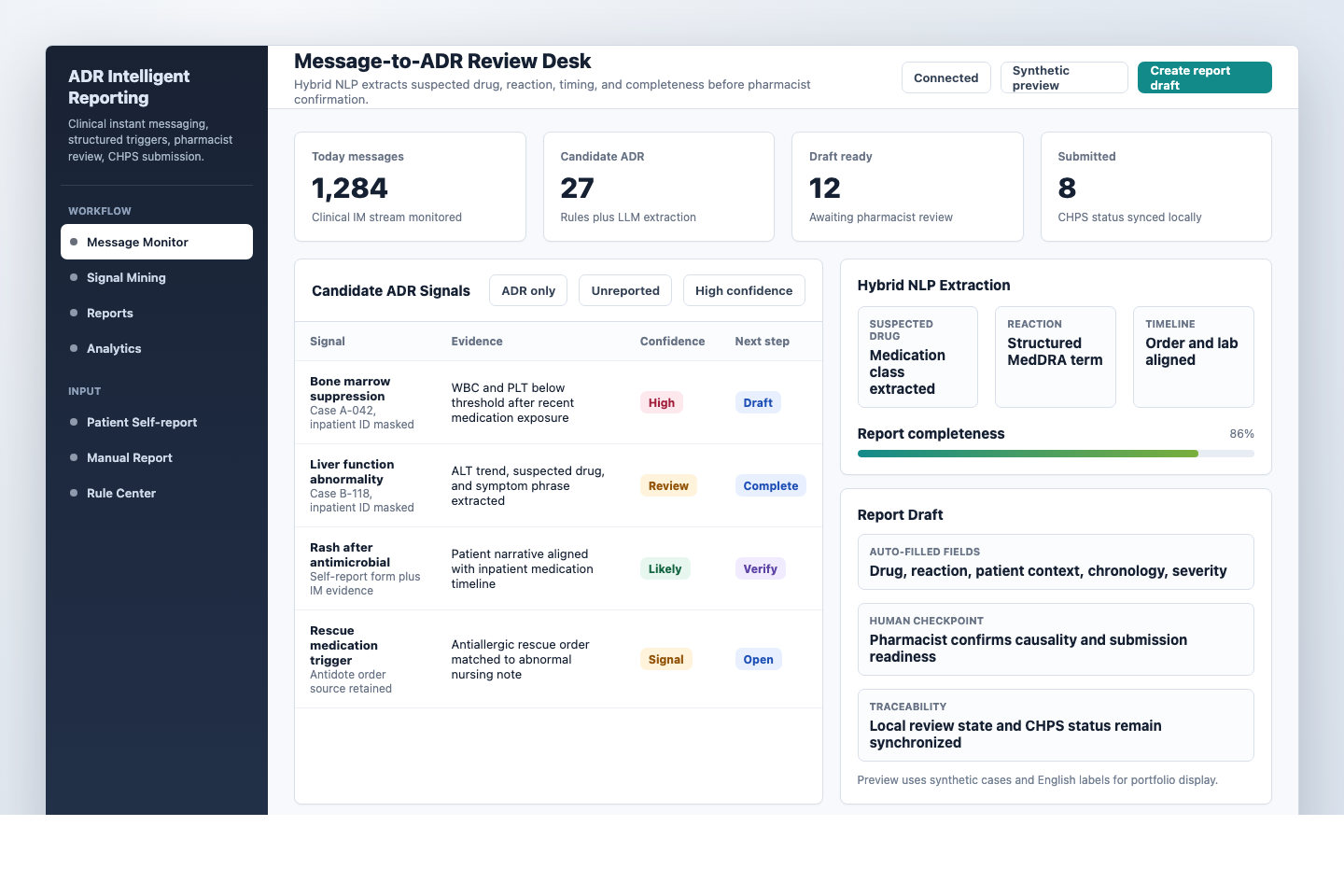
A medication-safety platform that turns clinical instant messages and hospital-data triggers into reviewable ADR signals, report drafts, and traceable submission workflows.
ADR evidence is often scattered across messages, laboratory abnormalities, medication orders, and patient narratives. Manual reporting is slow and easy to miss during routine clinical work.
A workflow for message monitoring, signal mining, patient self-report intake, report management, CHPS status synchronization, and rule-center configuration.
I designed the clinical workflow, data logic, review states, and hybrid NLP pipeline that keeps AI assistance subordinate to pharmacist confirmation.
A de-identified English preview generated from the system’s actual front-end structure.
The interface brings together message monitoring, rule-triggered safety signals, LLM-assisted extraction, and pharmacist review before report submission.
The value of the system is not only extraction accuracy; it is the translation of noisy clinical traces into a governed pharmacist workflow.
Clinical messages, medication orders, laboratory abnormalities, rescue-medication triggers, patient self-reports, and reporting-system status are normalized into the same review surface.
Rules protect high-value safety signals, while LLM-assisted extraction completes narrative fields and structures candidate ADR evidence for human review.
The system supports triage and drafting, but causality assessment, completeness judgment, and final submission remain pharmacist-led.
This platform shows implementation ability across clinical pharmacy, AI methods, and real hospital reporting constraints.
The work is connected to a 2026 SSRN preprint on zero-friction ADR reporting from clinical instant messaging using hybrid NLP.
The system is designed around practical interfaces and review queues rather than isolated model outputs, making it closer to deployable clinical infrastructure.
Conservative retention rules preserve high-value signals such as rescue medication and hematologic toxicity flags, reducing the risk of silent misses.